|
10/22/2023 0 Comments Idarucizumab dose 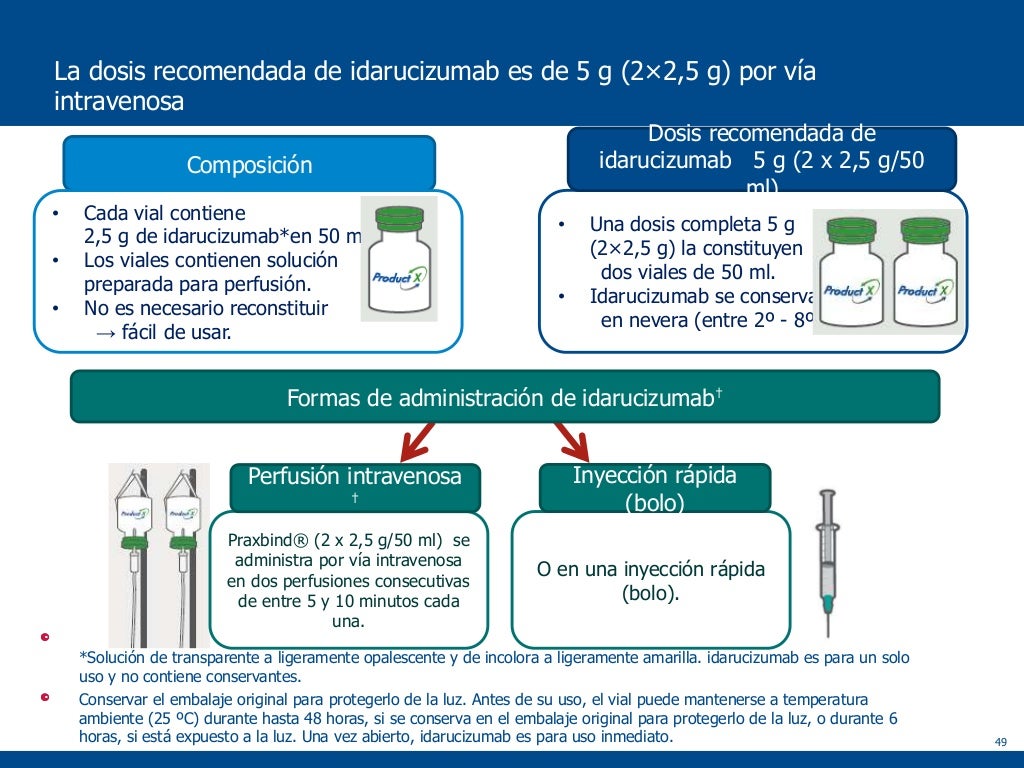
Each of these deaths could be attributed either as a complication of the index event or associated with comorbidities. 
The summary of product characteristics for idarucizumab states that during the ongoing RE‑VERSE AD study, 26 out of a total of 123 people died. Reversing the anticoagulant effect of dabigatran etexilate with idarucizumab exposes people to the thrombotic risk of their underlying disease restarting anticoagulant therapy should be considered as soon as is medically appropriate ( idarucizumab summary of product characteristics). Idarucizumab binds specifically to dabigatran and its metabolites and will not reverse the effects of any other anticoagulant. It is licensed for use in adults treated with dabigatran etexilate ( Pradaxa, Boehringer Ingelheim Limited) when rapid reversal of its anticoagulant effects is required for emergency surgery or urgent procedures, or in life‑threatening or uncontrolled bleeding. Regulatory status: Idarucizumab ( Praxbind, Boehringer Ingelheim Limited) was launched in the UK in December 2015. People may still need other supportive measures, for example blood products, to manage their bleeding and these should be considered as medically appropriate. In the interim analysis of an ongoing, phase III, uncontrolled, cohort study (RE‑VERSE AD n=90), treatment with a 5 g dose of idarucizumab completely reversed the anticoagulant effect of dabigatran etexilate in adults who had either serious bleeding or required urgent surgery. Its action is specific against the NOAC dabigatran etexilate. Idarucizumab is the first agent to be licensed in the UK that reverses the anticoagulant effect of a non‑vitamin K antagonist oral anticoagulant (NOAC). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
